PassaLab

Professor Daniele Passarella’s Research Group
Department of Chemistry
University of Milan
Check out our latest publications!
PROTAC-mediated degradation of peroxisomal d-aspartate oxidase: A novel strategy to modulate d-aspartate homeostasis for schizophrenia treatment

Eur. J. Med. Chem., 2026
Guided by STD-NMR, we designed a series of heterobifunctional degraders integrating a hDASPO-binding ligand (olanzapine) with established E3 ligase recruiters (lenalidomide or VH032–Me) and aliphatic linkers of varying lengths. The resulting bifunctional compounds were synthesized and evaluated in biochemical assays for their ability to bind hDASPO. Most compounds retained micromolar
inhibitory activity, indicating that derivatization at olanzapine position 10 preserves target engagement, with CRBN-recruiting analogues generally outperforming their VHL-based counterparts. Selected compounds were further investigated in cellular models, where PROTAC 17 demonstrated robust target engagement, positive cooperativity in ternary complex formation and dose-dependent degradation of hDASPO. Find out more here.
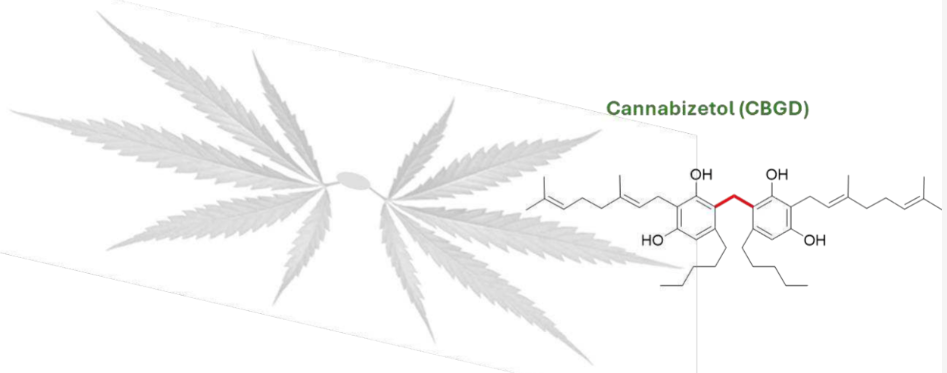
Cannabizetol, a Novel Cannabinoid: Chemical Synthesis, Anti- inflammatory Activity and Extraction from Cannabis sativa L
J. Nat. Prod., 2025
We report the first isolation of a previously unknown cannabinoid, cannabizetol (CBGD, 7), from Cannabis sativa extracts, representing the third member of the rare class of methylene-bridged dimeric cannabinoids.
Sustainable Synthesis of 1,2,3-Triazoles using Cyrene as a Biodegradable Solvent in Click Chemistry

ChemSusChem, 2025
The first successful synthesis of 1,2,3-triazoles using Cyrene as a biodegradable and non-toxic solvent in click chemistry has been developed. Find out more here.
PROTACs: Targeted Protein Degradation for Neurodegenerative and Cancer Targets
We are developing innovative PROTACs to degrade disease-related proteins implicated in neurodegenerative disorders (e.g. tau protein, DASPO) and cancers (tubulin). These bifunctional molecules hijack the ubiquitin-proteasome system to selectively remove otherwise undruggable targets.
Key publications: Pharmaceutics 2023, 15(5), 467; https://doi.org/10.3390/pharmaceutics15051467 BioRxiv 2025, https://www.biorxiv.org/content/10.1101/2025.05.22.655572v1

The project is coordinated by Prof. Passarella, member of the European research network ProteoCure – COST ACTION 20113.
Quantitative Live Cell Imaging of a Tauopathy model enables the Identification of a Polypharmacological Drug Candidate that Restores Physiological Microtubule Interaction
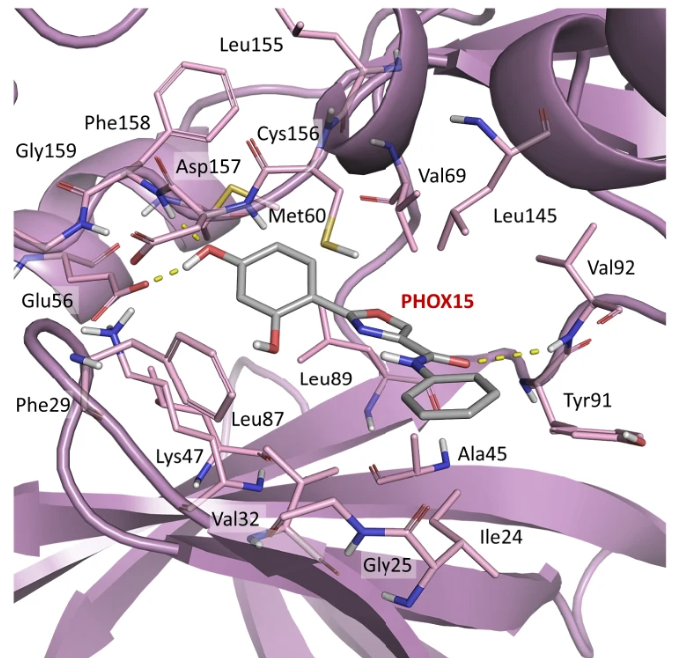
Nature Communications, 2024
A live-cell imaging of a tauopathy model enables screening of compounds that modulate taumicrotubule interaction and allows identification of a promising polypharmacological drug candidate that simultaneously inhibits tau aggregation and reduces tau phosphorylation. Find out more at the link.
A Green Approach to Nucleophilic Aromatic Substitutions of Nicotinic Esters in Cyrene

European Journal of Organic Chemistry, 2024
Cyrene has been used as an eco-friendly alternative to environmentally harmful polar aprotic solvents in SNAr reactions. This innovative protocol enabled the facile synthesis of a wide library of novel nicotinic esters, valuable precursors of bioactive compounds. This methodology could also be extended to scale-up processes, microwave-assisted synthesis, and one-pot telescoping reactions, thus enhancing its applicability in various eco-friendly synthetic contexts. Find out more at the link.
Stereodefined Synthesis of 3-Difluoromethyl-Benzoxaboroles: Novel Antimicrobials with Unlocked H-Bonding

Organic letters, 2026
Benzoxaboroles, prominent scaffolds in medicinal chemistry, are typically modified on the benzene ring. In contrast, functionalization of the oxaborol ring is less common and often challenging. Indeed, 3-hydroxy-benzoxaboroles are virtually impossible to isolate due to their tautomeric equilibrium with the carbonyl form. In this work, we introduce a novel class of stereodefined 3-difluoromethyl-benzoxaboroles. The replacement of the hydroxy group with −CHF2 preserves stability while promoting bioactivity, owing to the lipophilic H-bond donor properties of the latter. Find out more here.
Photochemical C4-Selective C–H Amination of Quinolines via N-Shift of Heteroaryl Azides

Organic letters, 2026
We report a photochemical strategy enabling the direct synthesis of 3,4-diaminoazines through C3-to-C4 nitrogen migration (N-shift). Blue LED irradiation of 3-azidoquinolines at room temperature allows C4-selective C–H amination while concurrently installing diverse amines at the C3–position. In contrast, N-shifts in 3-azidopyridines preferentially form diazepines, reflecting divergent reactivity governed by N-heterocycle electronics, as supported by computational analysis.
Synthesis of α-fluorocinnamate derivatives as novel cathepsin S inhibitors with in vitro antiproliferative activity against pancreatic cancer cells

Bioorg. Med. Chem., 2025
We have designed and synthesized three new derivatives basing on an isosteric replacement (H–F) at the level of cinnamate moiety. These derivatives emerged as potent covalent inhibitors of CatS (1.8–2.6 µM) with 2F showing also weak inhibition activity against CatL (20 %) and CatB (29 %). In vitro assays of 2F against pancreatic cancer cell lines BXPC3 and CAPAN1 revealed significant antiproliferative activity, with IC50 = 5.79 µM and 20.75 µM, respectively. To find out more details follow the page
Metal-Free, Selective Ortho-Deuteration of N-Heterocyclic Oxides

Adv. Synth. Cat., 2025
In this work, we present a metal-free, selective ortho-deuteration of N-heterocycles starting from their N-oxides, proceeding at room temperature in just 5 minutes. More about it at this link
Design, synthesis and in vitro validation of bivalent binders of SARS-CoV-2 spike protein: Obeticholic, betulinic and glycyrrhetinic acids as building blocks

Bioorg. Med. Chem., 2025
Bivalent compounds have been designed to simultaneously bind both pockets of the Spike protein, offering significant advantages over single molecules or the combination of the two natural products. In vitro cell assays using pseudotyped recombinant lentiviral particles with selected SARS-CoV-2 Spike proteins demonstrated that 1 and 2 exhibit enhanced activity in reducing viral entry into target cells compared to individual natural products. Check it out at this link
Nicotinic Acid Derivatives As Novel Noncompetitive α-Amylase and α-Glucosidase Inhibitors for Type 2 Diabetes Treatment

ACS Medicinal Chemistry Letters, 2024
A library of novel nicotinic acid derivatives, focusing on the modification of position 6 of the pyridine ring with (thio)ether functionalities, was mostly produced through an innovative green synthetic approach (Cyrene-based) and evaluated for their α-amylase and α-glucosidase inhibitory activity. Find out more under the Link
First Total Synthesis of Caerulomycin K: a Case Study on Selective, Multiple C–H Functionalizations of Pyridines
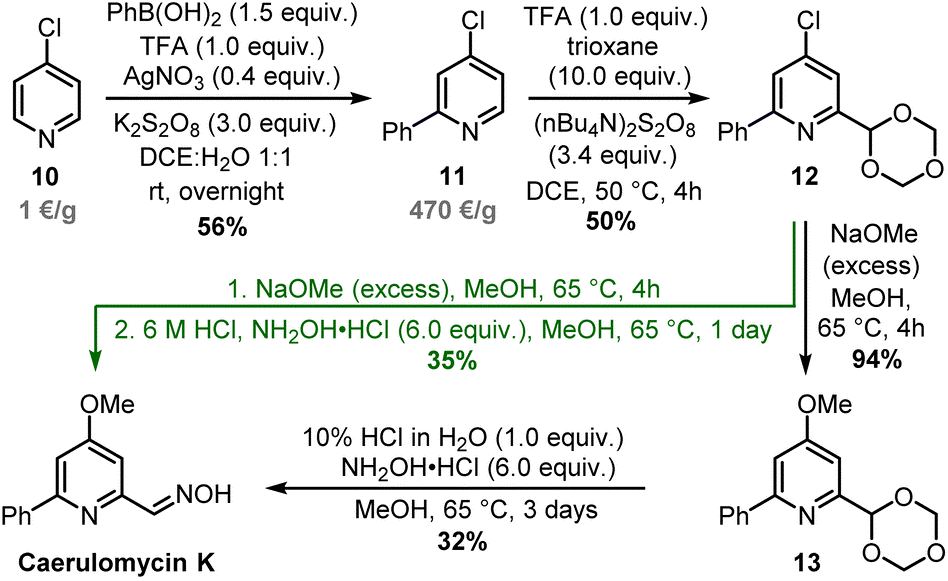
RSC Advances, 2024
Caerulomycins, natural alkaloids with antimicrobial properties, have been previously synthesized starting with highly pre-functionalized building blocks or requiring many functional group manipulations. In this work, we report the first total synthesis of caerulomycin K, a diversely trifunctionalized pyridine readily assembled in three steps exploiting the recent advancements in the C–H activation of N-heterocycles. Find out more under the Link
Rotational Motion in Bispidines: A Conformational Study
Organic Letters 2025, DOI: 10.1021/acs.orglett.5c02839
Francesco Migliano, Luca Pozzi, Andrea Citarella, Giovanni Macetti, Leonardo Lo Presti, Daniele Passarella* and Valerio Fasano*
Asymmetric Total Synthesis of (−)‐Glycybridin B, a Pharmacophore Screened Candidate for Tubulin Binding in Chemistry – A European Journal has the following publication status: Published Online
Chemistry – A European Journal 2025, DOI: 10.1002/chem.202502228
Alice Maiocchi, Maxim Shevelev, Zlata Boiarska, Juan Estévez-Gallego, Francesca Bonato, Paolo Orlando, Alessandra Chinosi, Emanuele Marcone, Andrea Citarella, Dragos Horvath, Michel O. Steinmetz, Andrea E. Prota, Alexandre Varnek, Valerio Fasano and Daniele Passarella
Structure-Based Discovery of Hsp90/HDAC6 Dual Inhibitors Targeting Aggressive Prostate Cancer
Journal of Medicinal Chemistry 2025, DOI: 10.1021/acs.jmedchem.5c00717
Andrea Citarella, Silvia Belluti, Davide Bonanni, Davide Moi, Isabella Piccinini, Arianna Rinaldi, Chiara Papulino, Rosaria Benedetti, Laura Cuoghi, Stefano Di Ciolo, Alessandra Silvani, Lucia Altucci, Luca Pinzi, Silvia Franchini, Daniele Passarella, Claudia Sorbi, Clelia Giannini, Carol Imbriano, Giulio Rastelli
Synthesis and multitarget inhibitory effect of indole-based ethyl cinnamate derivatives against SARS-CoV-2 Mpro and cathepsins for broad-spectrum anti-coronavirus activity
Bioorganic and Medicinal Chemistry 2025, DOI: 10.1016/j.bmc.2025.118258
Andrea Citarella, Giulia Sibille, Davide Moi, Alessandro Dimasi, Tommaso Braga, Lorenzo Dal Col, Lorenzo Ruberto, Stefano Pieraccini, Maurizio Sironi, Nicola Micale, Tanja Schirmeister, Valerio Fasano, Alessandra Silvani, Clelia Giannini, Giorgio Gribaudo, Daniele Passarella
Design, synthesis and characterization of aryl bis-guanyl hydrazones as RNA binders of C9orf72 G4C2 extended repeats
European Journal of Medicinal Chemistry, 2025, 293, 117736, DOI: 10.1016/j.ejmech.2025.117736
Alice Maiocchi, Martina Pedrini, Veronica Ferrari, Agata Sofia Assunçao Carreira, Vincenzo Maria D’Amore, Federica Santoro, Anna Di Porzio, Maddalena Bosetti, Riccardo Cristofani, Alessandra Silvani, Diego Brancaccio, Luciana Marinelli, Francesco Saverio Di Leva, Alessandro Provenzani, Angelo Poletti, Pierfausto Seneci
Automated fluorine-18 radiolabeling via an alkyne–azide cycloaddition reaction on a dual peptide-functionalized liposome surface for in vivo PET imaging
Frontiers in Pharmacology, 2025, 16, 1–16. DOI: 10.3389/fphar.2025.1566257
Marco Iannone, Marcelo Kravicz, Paolo Rainone, Antonia I. Antoniou, Stefano Stucchi, Silvia Valtorta, Arianna Amenta, Elia Anna Turolla, Sara Pellegrino, Daniele Passarella, Elisa Vino, Sergio Todde, Francesca Re, Pierfausto Seneci, Rosa Maria Moresco