Molecular Bases of Disease and Drug Design (MolBD )
We are the group of structural biology of the Institute of Biophysics - National Research Council, working in the buildings of the Dept. of Biosciences. Our aim is to dissect the molecular basis of human diseases. We exploit an integrated approach, blending structural biology techniques (such as X-ray crystallography, small angle X-ray scattering and cryo electron microscopy) with protein biochemistry/biophysics and computational biology.
Once the underlying pathological mechanism is well described, the same structural and biochemical tools are employed to screen and design novel drugs.
We are involved in several collaborative projects both with academic groups and pharmaceutical companies.
3
CNR Researcher
eloise.mastrangelo@unimi.it eloise.mastrangelo@cnr.it

Francesco Boni
PhD Student
francesco.boni@mi.ibf.cnr.it
francesco.boni@unimi.it
Elisa Fagnani
Post-graduate fellow
elisa.fagnani@ibf.cnr.it

Doris Mirdita
Molecular Biotechnology and Bioinformatics Student

Scientific Projects
GSN
BIR
Dengue RNA pol
AIF
SAVIOR
ASIC
GCAP
Gelsolin physiopathology (GSN)
Fondazione Telethon Italia - exploratory project GEP15070. 03/2016-05/2017
Amyloidosis Foundation: Research Grant 02/2018 - 01/2019
PI: Matteo de Rosa. Staff: Francesco Boni, Eloise Mastrangelo, Mario Milani (CNR-IBF), Riccardo Porcari, Guglielmo Verona (UCL, Londra), Alberto Barbiroli (UNIMI), Luisa Diomede, Margherita Romeo (Istituto 'Mario Negri', Milano)
Gelsolin is the prototype of a superfamily of Ca-dependent proteins and it is responsible for the assembly, disassembly and scavenging of actin by means of its severing and capping activities. Due to its pivotal physiological activities and the ubiquity nature, gelsolin plays a main role in a plethora of physiological processes, such as cell motility and division, organelle trafficking and muscle contraction. Moreover, increasing evidence suggests a central role in cell metabolism and signalling in an actin-independent fashion. As a consequence, alteration in gelsolin expression levels or deregulation of its activities have been observed in several diseases, cancers in particular. While the role of gelsolin in these pathological states is still to be elucidated, the protein is directly responsible for a rare amyloidosis due to toxic gain-of-function mutations. Integrating structural, biochemical and biophysical techniques, the GSN project aims at elucidating the molecular bases of gelsolin-related diseases and at providing a structural description of gelsolin physiological activities. In parallel, we recently started an in silico screening of gelsolin stabilizers to develop novel drugs against gelsolin-related diseases.
Type I BIR domain inhibitors in cancer therapy: Searching drugs for regulation of the NF-kB pathway
Associazione Italiana per la Ricerca sul Cancro (AIRC-MFAG, Italy)
2016-01 to 2019-03 | Grant: 17083
The over-expression of Inhibitors of apoptosis proteins (IAPs) enhances cell survival and resistance to anticancer agents. IAPs are E3-ligases, ubiquitylating substrates for the regulation of NF-kB; furthermore, they sequester caspases to prevent apoptosis. IAPs interactions occur through type-I and type-II BIR (Baculovirus IAP repeat) domains. Smac-mimetics (SMs), mimicking the N-terminal peptide of the natural antagonist of IAPs, interact with type-II BIRs, relieving caspases from X-linked IAP (XIAP) and leading to cellular IAPs (cIAPs) proteasomal degradation. Although SMs are currently validated candidates for cancer therapy, some cancer-cell line retains SM-resistance due to cIAP2 overexpression and NF-kB re-activation.
IAPs-mediated regulation of NF-kB signaling is based on the formation of different protein complexes by type-I BIRs, regulating ubiquitin-dependent signal transduction. We analyzed X- and cIAP-BIR1 surfaces to identify hot-spots involved in protein-protein interactions. Virtual docking selected hits able to impair BIR1-based complexes with predicted low micromolar affinities (i.e.NF023) that were experimentally confirmed. 3D-structural analysis of the protein-ligand complexes obtained allowed the optimization of specific and selective drug candidates. Treatment of cancer-cells with the selected compounds will validate their effects on IAPs-dependent signaling cascades. This approach represents a novel strategy to promote apoptosis in cancer, providing new insights on the regulation of NF-kB pathway.
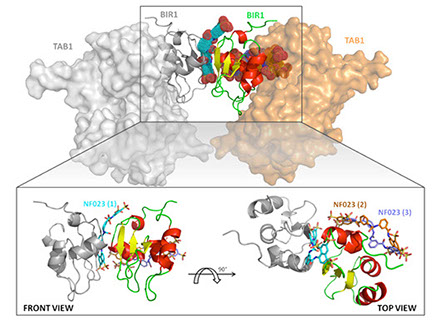
PI: Eloise Mastrangelo. Staff: Federica Cossu (Senior Scientist), Doris Mirdita (master degree student). Collaborations: Daniele Lecis (Istituto Tumori).
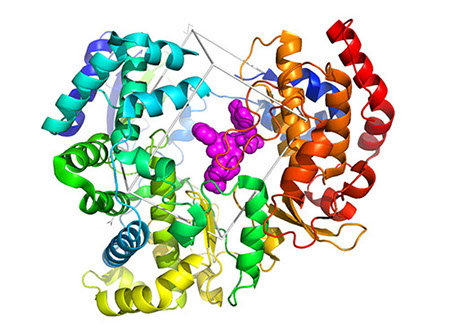
Over the past years, many RNA viruses were identified or 're-discovered', including life-threatening flaviviruses, such as ZIKA and the four Dengue virus serotypes (DENV1-4). Thus, there is a pressing need for new therapeutics, since those currently available are scarcely effective. Target-based design of inhibitors of flaviviral enzymes may be a fruitful strategy for the development of inhibitors of flavivirus replication. Between them, the NS5 C-terminal RNA-dependent RNA polymerase (RdRp) plays an essential role in the amplification of subgenomic RNA. Since human cells lack RdRp, this class of enzymes appears as one of the most promising targets for antivirals development. In this context, in a thorough search for new anti-DENV compounds, we performed an in silico docking search targeting a region around the active site of DENV3 RdRp using a focused-library of molecules produced by the University of Perugia. Among the in silico predicted inhibitors, one heterotricyclic compound, named UP4, was able to inhibit not only DENV3 RdRp activity in vitro, but also several flaviviruses in cell cultures, with 2-10 micromolar IC50 values. In this project, we use protein crystallography to characterize the structure of the hit/enzyme complex in order to perform rational structure-based modifications of UP4.
Anti-viral drug discovery strategies: structure-based development of Dengue virus RNA dependent RNA polymerase inhibitors
MIUR (SIR, Italy) 2015-06 to 2018-05 | Grant RBSI14C78S
We plan to synthesize a series of hit analogs, aiming at more potent and selective RdRp inhibitors, and achieve a structure-activity relationship characterization of this class of new compounds. Furthermore, since the structure of the RdRp/ssRNA initiation complex is not available for a flavivirus, we aim to elucidate (through a combination of 3D structural data, point mutation studies, activity assays) the mechanism of de novo RNA synthesis. We believe that the combined approaches described above, supported by our key preliminary results, will provide unique information not only for the design of future therapeutics, but also for basic virology.
PI: Eloise Mastrangelo. Staff: Mario Milani (Senior Scientist), Matteo de Rosa (Senior Scientist). Collaborations: Gilles Querat (University of Marseille), Violetta Cecchetti, Giuseppe Manfroni, Rolando Cannalire (University of Perugia).
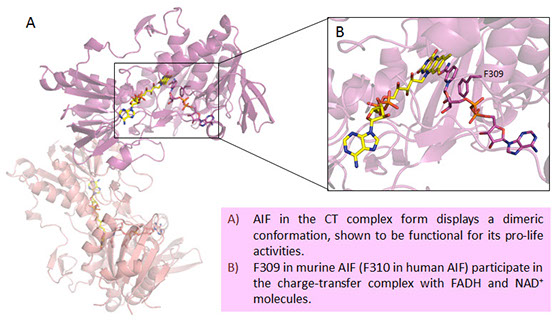
We aim to increase understanding of AIF molecular properties and details of its interaction with CHCHD4 and to reveal how they are altered in AIFM1 pathogenic variants, also in collaboration with cellular biochemists and biologists specialized in neurodegeneration.
We expect to define a solid rationale based on the combination of expertise and techniques, implementing a virtuous cycle of structural, biochemical and cell-based experiments to give a comprehensive framework of AIF roles in mitochondria. On the medium long-term our strategies and results will:
-Give instruments to study further overcoming AIF defective forms;
-Set the bases for therapeutic intervention for the treatment of AIF-based forms of neurodegeneration, hopefully with the support of SMEs attracted by our disseminated research.
Structural and biochemical bases of the pathogenicity of the Apoptosis Inducing Factor (AIF)
AIF is a mitochondrial intermembrane component that fulfills the double role of apoptotic signal in a caspase-independent manner (pro-death function) and of flavoprotein required for mitochondrial biogenesis and maintenance, involved in the formation of the respiratory chain complexes (pro-life function). Since 2010, an increasing number of mutations in AIFM1, the gene encoding AIF, have been identified as the cause of human neurodegenerative mitochondriopathies. This pathology reflects in neural disorders mainly leading to severe encephalopathies and denervation, seizures, blindness and deafness, associated with oxidative phosphorylation (OXPHOS) deficiency. Beyond to NADH-oxidation, it has been stressed the role of AIF in the import and folding of other mitochondrial proteins directed to the mitochondrial intermembrane space, mainly via its interaction with CHCHD4 (Coiled-coil-helix-coiled-coil-helix domain-containing protein 4).
PI: Federica Cossu Team: Eloise Mastrangelo, Luca Sorrentino, Paolo Cocomazzi. Collaborations: Alessandro Aliverti (University of Milano)
SAVIOR Project: Selecting AntiViral Inhibitors Of Rotavirus
FIRB-Futuro in Ricerca 2013 project (Grant RBFR13209E Title "SAVIOR Project: Selecting AntiViral Inhibitors Of Rotavirus")
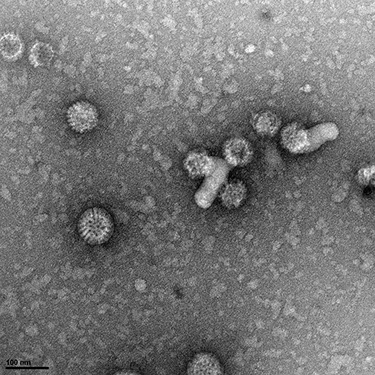
In developing countries Rotavirus gastroenteritis is responsible for about 452,000 infant deaths annually, despite the commercial availability since 2006 of two attenuated vaccines. Currently, there are no specific antiviral therapies available. The SAVIOR project proposes a multidisciplinary approach to identify molecules able to inhibit the early stages of Rotavirus replication. We aim at identifying molecules interfering with the viral replication machinery and with specific cellular proteins already discovered (by a genome-wide RNAi screening) as essential for viral replication, but dispensable for cell survival. We recently identified a compound able to disrupt viroplasms and therefore compromise their function, thus impairing Rotavirus replication.
PI: Michela Bollati. Staff: Eloise Mastrangelo (Group Leader), Mario Milani (Gruop Leader), Matteo de Rosa (Senior Scientist).
Acid- Sensing Ion Channel 1a (ASIC1a): a promising target in neurological disorders
Extracellular acidosis affects several neuroinflammatory/neurodegenerative disorders. Under physiological conditions, the extracellular pH levels in brain are maintained at 7.4 through various protons (H+) transporting mechanisms. However, during inflammation such levels are significantly reduced causing a long lasting acidosis in brain. Tissue acidosis is a common feature in pathological conditions such as multiple sclerosis (MS), stroke and epilepsy, which are characterised by massive cell loss, essentially leading to deterioration in quality and function of tissues. Several ion channels modulate this reduction of pH levels included the Acid-Sensing Ion Channels (ASICs). Among ASICs, ASIC1a is the major isoform, characterized by broad pH-detecting variation, cation permeability and high expression levels in the neurons of the CNS. It is known that pharmacological inhibition of ASIC channels exerts beneficial effects, improves clinical outcomes and protects axons from degeneration. Nevertheless, ASIC inhibitors so far available are not very specific nor particularly efficient, limiting substantially both the study of its physiological role and its validation as therapeutical target. Main aim of the project is to determine the ASIC1a structural and molecular features needed to design potent, isoform-selective and brain-penetrant analogs of the currently available drugs. The molecular mechanisms acting downstream of ASICs are far from being elucidated. Therefore, selectively targeting ASIC1 is crucial to clarify the role of ASIC isoforms in neurodegenarative disorders.
PI: Mario Milani. Staff: Eloise Mastrangelo. Collaborations: Andrea Menegon and Luca Muzio (San Raeffele hospital, Italy), Pierfausto Seneci (Univ. Milano, Italy)
Cone dystrophies and retinal degeneration from protein structures to biological networks. Toward the design of therapeutic molecules
Supported by Fondazione Telethon grant GGP16010 (02/2017-30/2020)
PI: Mario Milani. Staff: Francesco Boni, Eloise Mastrangelo, Collaborations: Daniele Dell'Orco (Univ. of Verona. Italy), Lorenzo Cangiano (Univ. of Pisa, Italy)
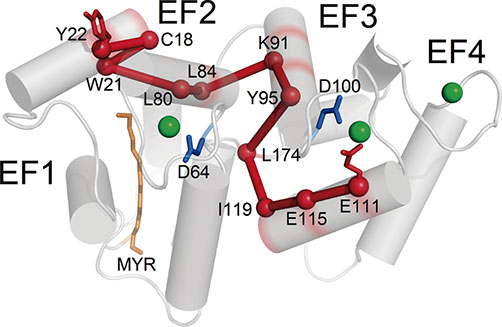
In patients suffering from autosomal dominant cone distrophy (COD), 14 mutations have been found in the GUCA1A gene coding for the guanylate cyclase activating protein 1 (GCAP1), an essential component of the phototransduction cascade. COD-GCAP1 mutants exhibit an impaired Ca2+-sensitivity and constitutively activate guanylate cyclase (GC) targets, thus deregulating the cGMP metabolism and increasing the influx of Ca2+ through the cyclic nucleotide-gated(CNG)-channels. Unraveling the effects of COD mutations on GCAP1 structure/ function and their roles in the perturbation of the complex with the target GC will constitute the basis for the design of specific molecules or peptides capable of interfering with the abnormal regulation of GC. An integrated experimental and computational approach is applied to: i) characterize the molecular basis of disease-onset from single proteins to complexes and networks; ii) quantify the perturbed homeostasis of calcium and cGMP; iii) identify molecules able to restore the normal homeostasis of second messengers.
The Structural Biology Group comprises members from both the DBS-UNIMI and the IBF-CNR. The content herein is not regulated by the University of Milan.