Apis group Amino acids, Peptidomimetics, peptides, protein InteractionS group
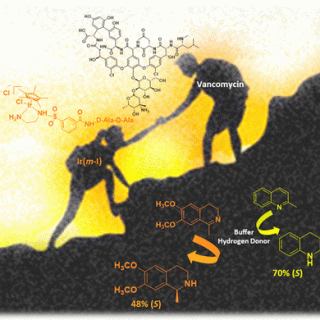
Based on the supramolecular interaction between vancomycin (Van), an antibiotic glycopeptide, and D-Ala-D-Ala (DADA) dipeptides, a novel class of artificial metalloenzymes was synthesized and characterized. The presence of an iridium(III) ligand at the N-terminus...
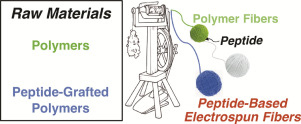
Nanofiber films produced by electrospinning currently provide a promising platform for different appli- cations. Although nonfunctionalized nanofiber films from natural or synthetic polymers are extensively used, electrospun materials combined with peptides are gaining more...
Bioinspired smart materials represent a tremendously growing research field and the obtainment of new building blocks is at the molecular basis of this technology progress. In this work, colloidal materials have been prepared in...
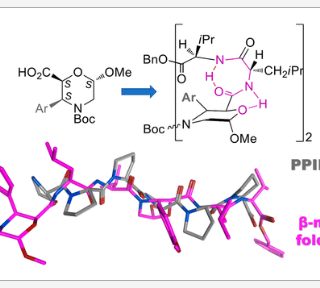
A new non-natural β-amino acid, named 3-Ar-β-Morph, was designed and synthesized via a regio- and diastereoselective Pdcatalyzed C(sp3)H-arylation of the corresponding 2S,6S-(6-methoxymorpholin-2-yl)carboxylic acid, readily available from glucose. According to the computational prevision and confirmed...
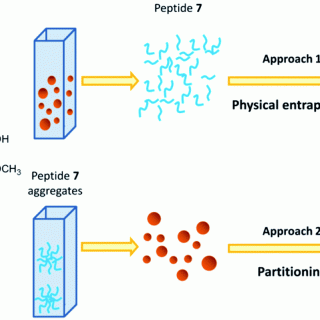
The exploitation of self-assembled systems to improve the solubility of drugs is getting more and more attention. Among the different types of self-assembled biomaterials, peptides and in particular peptides containing non-coded amino acids (NCAPs)...
The outer membrane (OM) of Gram-negative bacteria is a highly selective permeability barrier due to its asymmetric structure with lipopolysaccharide (LPS) in the outer leaflet. In Escherichia coli, LPS is transported to the cell...
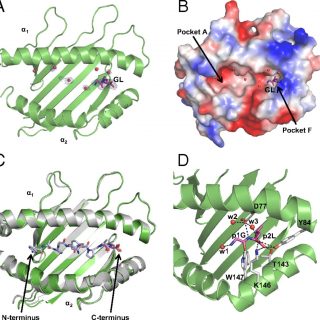
We have determined a suite of crystal structures of MHC-I molecules that provide insights in the molecular basis underlying peptide binding and selection by tapasin. We demonstrate that the scoop loop of tapasin binds...
The chiral structure of antibiotic vancomycin (Van) was exploited as an innovative coordination sphere for the preparation of an IrCp* based hybrid catalysts. We found that Van is able to coordinate iridium (Ir(III)) and...
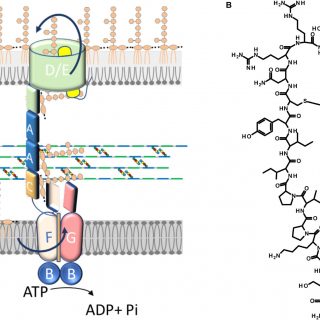
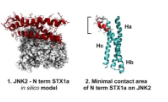
The neuronal loss caused by excessive glutamate release, or ‘excitotoxicity’, leads to several pathological conditions, including cerebral ischemia, epilepsy, and neurodegenerative diseases. Over-stimulation of presynaptic N-methyl-D-aspartate (NMDA) receptors is known to trigger and support...
The human inducible phospho-fructokinase bisphosphatase isoform 3, PFKFB3, is a crucial regulatory node in the cellular metabolism. The enzyme is an important modulator regulating the intracellular fructose-2,6-bisphosphate level. PFKFB3 is a bifunctional enzyme with...








comments